ReBoost Nasal Spray Recalled for Microbial Contamination
This recall was posted 5 months ago. Check the FDA source for the latest status.
Class I Recall — Most Serious
This is a Class I recall — the most serious type. Products covered by this recall may cause serious health consequences or death.

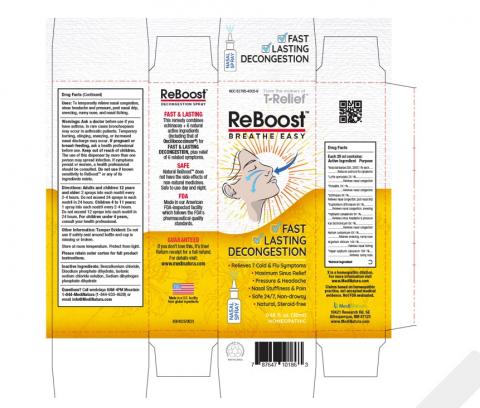
MediNatura New Mexico, Inc. has announced a voluntary recall of one lot of ReBoost Nasal Spray due to microbial contamination. This recall affects consumers nationwide.
Products Affected:
- ReBoost Nasal Spray, 0.68 fl oz
- Lot Number: 224268
- Expiration Date: 12/2027
- NDC: 62795-4005-9
- UPC: 787647 10186 3
The product is packaged in a 20mL bottle within a white and yellow carton and is used to temporarily relieve nasal congestion and related symptoms.
Reason for Recall:
The recall was issued because the nasal spray was found to contain yeast, mold, and a microbial species called Achromobacter at levels exceeding safety specifications. This contamination poses a risk of life-threatening infections, particularly for individuals with weakened immune systems.
What Consumers Should Do:
- Immediately stop using the recalled ReBoost Nasal Spray.
- Consumers who purchased directly from MediNatura should contact them at [email protected] for a refund.
- Those who bought the product from a retailer should return it to the place of purchase.
- Contact a healthcare provider if you experience any health issues related to this product.
To date, no adverse events have been reported. Consumers can report any adverse reactions to the FDA's MedWatch program online or by mail or fax.
For more information, contact MediNatura New Mexico, Inc. at (800) 621-7644 or [email protected], Monday through Friday, 8:00 AM to 5:00 PM MST.
Where Products Were Sold
This recall affects products distributed nationwide across the United States.