TRUE METRIX Glucose Meters: Label Update for E-5 Error
This recall was posted 3 months ago. Check the FDA source for the latest status.
Class I Recall — Most Serious
This is a Class I recall — the most serious type. Products covered by this recall may cause serious health consequences or death.
Reported Health Impact
114
Illnesses
1
Death
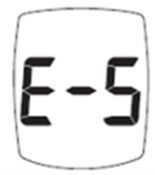
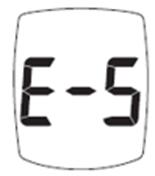
Trividia Health, Inc. has announced a labeling update for all TRUE METRIX Blood Glucose Monitoring Systems due to a critical instruction regarding the E-5 error code. This update affects devices distributed in the U.S., U.K., Mexico, Australia, and the Caribbean.
Affected Products:
- TRUE METRIX
- TRUE METRIX AIR
- TRUE METRIX GO
- TRUE METRIX PRO
Reason for Recall:
The current instructions do not emphasize the need for immediate medical attention if users experience symptoms of high glucose and receive an E-5 error code. This error indicates a very high blood glucose level (> 600 mg/dL) or a test strip error. Delayed treatment could lead to serious health consequences or death.
What Consumers Should Do:
- Retest with a new test strip if an E-5 error appears.
- If symptoms such as fatigue, excess urination, thirst, or blurry vision occur, seek medical attention immediately.
- If no symptoms are present, retest with a new strip. If the error persists, contact Trividia Health at 1-800-803-6025.
Health Risks:
Since 2014, there have been 114 reported serious injuries and one death linked to the E-5 error code.
For further information, contact Trividia Health Customer Care at 1-888-835-2723 or visit their website. Consumers can continue using the meters; no product returns or replacements are necessary. Report any adverse reactions to the FDA's MedWatch Program.
Affected Products
| Product Name |
|---|
| Blood Glucose Monitoring Systems |
| Blood Glucose Monitoring Systems |
Where Products Were Sold
This recall affects products distributed nationwide across the United States.
Contact Information
Media Contact
Annmarie Ramos